Immunotherapy

How is the immune system defined?
The lymph glands, spleen, and white blood cells comprise the immune system. It safeguards the body against infection, illness, and disease, including cancer. However, cancer can develop when the immune system recognizes cancer cells but cannot destroy them or when the cancer cells conceal themselves from the immune system.
Immunotherapy
Immunotherapy is a biological therapy that bolsters the body's natural defences against cancer cells and infections. Conventional cancer treatment is associated with a high rate of adverse events, as it does not distinguish cancer cells from healthy cells. Immunotherapy, for example, has overcome this obstacle by training the patient's natural immune system to recognize and selectively kill cancer cells, resulting in a reduced number of manageable general side effects.
Your physician may prescribe immunotherapy alone or in combination with other cancer treatments, depending on the factors listed below for each patient:

Immunotherapy indications/applications
Immunotherapies have been approved and are currently being used to treat a variety of cancers worldwide, including the following:
There are numerous other cancers for which immunotherapy research is ongoing.
Types of immunotherapy

1) Inhibition of immune checkpoints
Checkpoint inhibitors are an immunotherapy class of medication. They work by inhibiting the immune system's ability to attack cancer cells.
By attacking bacteria and viruses, our immune system protects us from disease. A T-cell is one of the most common types of immune cell that performs this function.
T-cells contain proteins that activate an immune response (cause it to become active) and proteins that inhibit it (become inactive). These proteins are referred to as checkpoint proteins.
When the checkpoint proteins are activated, T-cells can attack and destroy other cells, such as cancer cells. On the other hand, T-cells can begin to destroy healthy cells and tissues if they remain active for an extended period or if they react to things they should not. This is why T cells also have additional checkpoint proteins that instruct them when to rest and refrain from attacking.
Certain cancer cells produce large amounts of proteins that inhibit T cells and prevent the immune system from attacking and destroying cancer cells.
Checkpoint inhibitors are drugs that prevent checkpoint proteins from being activated by cancer cell proteins. By inhibiting these signals, checkpoint inhibitors prevent T cells from turning off, allowing them to locate and attack cancer cells.
The following is a list of FDA-approved immunotherapies:

2) T-cell therapy with adoptive cells/chimeric antigen receptor (CAR) T-cell therapy
Adoptive cell transfer is a type of immunotherapy in which the patient's T cells are used to aid in the fight against cancer. T cells are isolated from the patient's blood or tumor and treated in the laboratory with substances that enhance their ability to recognize and kill cancer cells in their bodies. Numerous types of adoptive cell transfer therapies have been developed, but the one that has made the most clinical progress to date is called chimeric antigen receptor (CAR) T-cell therapy. Apheresis is used to collect a patient's own T cells for CAR T-cell therapy.
Blood is drawn from one of the patient's large veins and circulated through an apheresis machine, which separates the blood's constituents. Only the T cells are removed from this blood, and the remaining blood is returned to the patient. T cells are sent to a laboratory to be genetically engineered to produce receptors called chimeric antigen receptors on their surfaces (CARs). These unique receptors enable T cells to recognize and associate with a specific antigen expressed on tumor cells. (The most advanced CAR T-cell therapies target an antigen found on B cells called CD19.) Once modified, the T cells are multiplied into hundreds of millions of cells in the laboratory. These newly developed CAR T cells are dubbed "attacker" cells. When sufficient cells are obtained, they are frozen and transported to the hospital where the patient is being treated.
Before receiving the CAR T cells, the patient receives chemotherapy to help the body prepare for the impending CAR T cells. Once infused into the patient's bloodstream, the CAR T cells multiply and begin recognizing and attacking cancer cells that express the targeted antigen. CAR T-cells may remain in the body for months after the infusion is complete, resulting in long-term remissions in some patients with blood cancer. CAR-T cells are currently approved for use in young patients with relapsed B-cell acute leukemia and relapsed lymphomas.
The FDA has approved two CAR T-cell therapies.

3) Monoclonal Antibodies
The immune system combats foreign invaders in part by producing billions of distinct antibodies. Antibodies are proteins that bind to antigens (a substance from the environment, such as bacteria or viruses, or inside the body, such as a cancer cell). Antibodies circulate throughout the body until they contact and attach to a specific antigen for which the antibody has receptors. Once attached, the antibody can recruit other components of the immune system to assist in the destruction of foreign cells containing the offending antigen.
To treat cancer, researchers can create antibodies in the laboratory specific to a particular antigen, such as those found most frequently on cancer cells. Possessing the ability to identify and target such antigens would result in the least amount of damage to normal cells. Monoclonal antibodies "mark" cancer cells, making them more visible and susceptible to destruction by the immune system. Monoclonal antibodies act as target-seeking missiles, locating and attaching to tumor-specific antigens before delivering the toxic substance directly into the cancer cell.
Monoclonal antibodies are classified into three types:
Naked monoclonal antibodies- These treatments are self-contained. They are not tainted with drugs or radioactive material. While the majority of naked monoclonal antibodies bind to antigens on cancer cells, some also bind to antigens on non-cancerous cells.
The following are examples of naked monoclonal antibodies.
Bispecific monoclonal antibodies-These treatments combine two distinct monoclonal antibodies that can bind to two distinct proteins simultaneously.
Conjugated monoclonal antibodies- Monoclonal antibodies that have been conjugated. These medications havea chemotherapeutic agent or a radioactive materialpertaining to them. They are used to administer medication to thetumour cells. The following is a list of conjugated monoclonal antibodies:
4) Vaccines for therapeutic purposes

Certain types of blood cancer are being treated experimentally with vaccines. Cancer vaccines teach the immune system to recognize and defend against cancer cells. These vaccines are intended to slow or stop cancer cell growth,prevent recurrence of cancer that has been treated and eliminate cancer cells that have not been destroyed by treatment. Cancer vaccines expose the body's immune system to cancer-associated molecules, enabling it to destroy them. Researchers are still concentrating their efforts on developing vaccines against blood cancers.Immunotherapy will be administered in several different ways.Typically, we will administer the treatment to you via a drip into your bloodstream.
What are the side effects of immunotherapy?
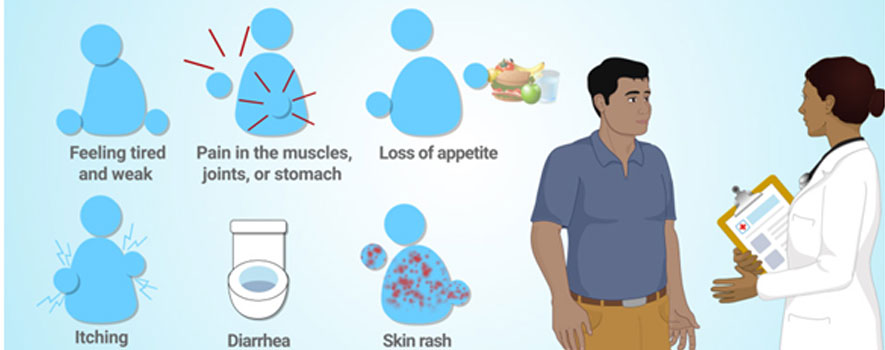
Immunotherapy can have a range of side effects ranging from mild to severe. Fatigue and flu-like symptoms, such as fever and chills, headaches, and muscle and joint pain, are the most common.
Almost everyone who undergoes immunotherapy initially experiences these side effects. It usually improves as your treatment progresses. These symptoms begin between two and four hours after your injection and last approximately 12 hours. Paracetamol can assist in alleviating these symptoms.
Immunotherapy may also result in a temporary decrease in red, white, or platelet blood cells. A decrease in white blood cells can increase one's susceptibility to infection. Reduced red blood cell counts can result in fatigue and shortness of breath. If the levels fall too low, you may require a blood transfusion. A decrease in blood platelets can result in nosebleeds, bleeding gums after brushing your teeth, or an abundance of small red spots or bruises on your arms and legs called petechia. Additionally, itchy, dry skin or rashes may occur.
If you experience any of these side effects, you should contact your health care provider. Your physician will monitor your blood counts regularly.
Additionally, nausea and vomiting, diarrhea, loss of appetite, and changes in taste or a metallic taste in the mouth are possible symptoms. Additionally, you may experience a sore throat and difficulty swallowing. Because these symptoms may indicate dehydration, weight loss, or malnutrition, it is essential to notify your doctor or nurse if they persist.
You may experience depression, anxiety, or difficulty sleeping during treatment. These sensations may be a side effect of immunotherapy, or they may be a reaction to your diagnosis. You can request psychological support from your medical team if you feel the need for someone to talk to.
